
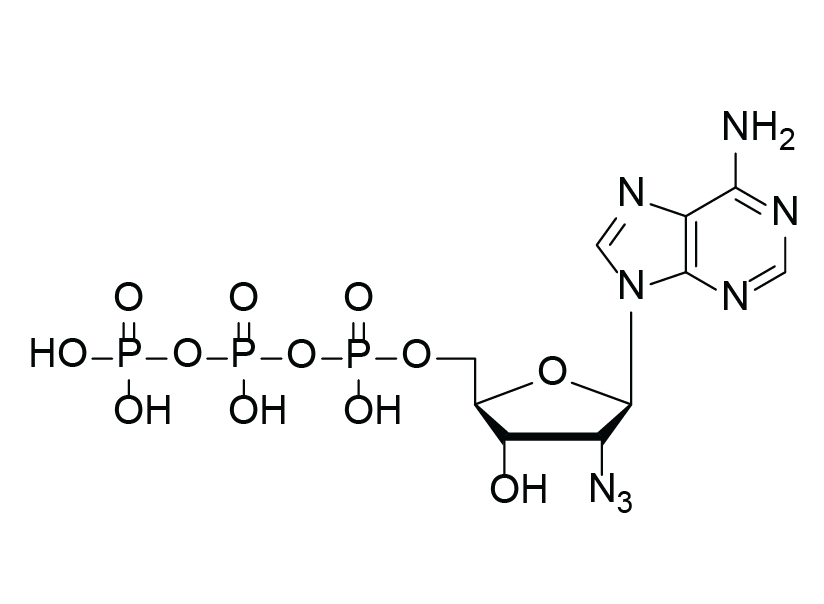
2′-Azido-2′-dATP
Triphosphate for modifying of 3'END RNA
| Size | Catalog No. | Price |
|---|---|---|
| 1 µmol | BCT-34-S | € 250,00 |
| 5 µmol | BCT-34-L | € 920,00 |
Chemical Properties
-
Molecular Formula
C10H15N8O12P3
-
Shelf Life
12 months unopened after receipt
-
Storage Conditions
-20 °C, dark, dry
-
Molecular Weight
532.19 g/mol (free acid)
-
Purity
≥ 95% (HPLC)
-
Physical State
colorless solution (100 mM, pH 7.5)
-
CAS Number
69093-69-2
-
Absorption (max)
λmax = 258 nm
-
Ɛ (max)
15.400 cm-1M-1
Product Information
A Nucleotide Analogue for the Selective 3´End-Labeling
Molecular Structure and Applications of 2′-Azido-2′-dATP
2′-Azido-2′-deoxyadenosine 5´-triphosphate (2´-Azido-2´-dATP) is a chemically modified analogue of adenosine triphosphate (ATP), with an azido (N₃) group replacing the 2′ hydroxyl (OH) group of the sugar backbone.
2′-Azido-2′-dATP is designed for the site-specific, post-synthetic introduction of one to five azido groups at the 3′ end of nucleic acids, using enzymes such as poly(A) polymerase or terminal deoxynucleotidyl transferase (TdT). These azido groups enable the 3′ end to be modified with fluorescent dyes, biotins, linkers, and cell-targeting ligands via click chemistry.
2′-Azido-2′-dATP is also ideal for generating internally modified RNAs. When added to an in vitro transcription (IVT) reaction mixture, RNA polymerases efficiently incorporate 2′-azido-2′-dATP into the nascent RNA strand. These RNA transcripts can then undergo further modification with high specificity and sensitivity using bioorthogonal click chemistry.
The azido group: Biorthogonal RNA Labeling via Click Chemistry
The azido group is a biorthogonal functionality, meaning that it does not interfere with natural biomolecules. The azido group reacts selectively with:
- alkynes, forming triazoles via copper-catalysed azide–alkyne cycloaddition (CuAAC) or copper-free strain-promoted azide–alkyne cycloaddition (SPAAC).
- phosphines, forming amides via Staudinger ligation.
Both reactions fulfill the criteria for click chemistry, enabling the highly efficient ligation of labels as fluorescent dyes, biotins, linkers, or cell-targeting ligands to nucleic acids. The development of the azide–alkyne cycloaddition was recognized with the Nobel Prize in Chemistry in 2022.
Application Areas for 2′-Azido-2′-dATP
- 3′-End Labeling of Nucleic Acids: post-transcriptional enzymatic incorporation / addition to the 3´-end, followed by click conjugation with dyes, biotin, or ligands.
- Internal Labeling of RNA: co-transcriptional incorporation into RNA by in vitro transcription (IVT), followed by click conjugation with dyes, biotin, or ligands.
- Advanced Bioconjugation: allows the attachment of sugars or targeting moieties for active mRNA transport into cells.
- Copper-Free Click Chemistry In Vivo: the azido group of 2′-Azido-2′-dATP is a biorthogonal functionality, meaning that it does not interfere with natural biomolecules.
- Compatible with Enzymatic Incorporation: 2′-azido-2′-dATP is accepted as substrate by T7 RNA polymerase, poly(A) polymerase, and TdT.
Protocols and Resources
baseclick provides detailed protocols for:
- 3′-End modification of nucleic acids using 2′-azido-2′-dATP
- Click conjugation with fluorescent dyes, biotin, linkers, and targeting ligands
LITERATURE
Bioorthogonal mRNA labeling at the poly (A) tail for imaging localization and dynamics in live zebrafish embryos, K. J. Westerich et al., 2020, Chemical Science, Vol. 11(11), p. 3089-3095.
https://doi.org/10.1039/C9SC05981D
Enzymatic Generation of Double‐Modified AdoMet Analogues and Their Application in Cascade Reactions with Different Methyltransferases, M. Erguven et al., 2022, ChemBioChem, Vol. 23(24), e202200511.
https://doi.org/10.1002/cbic.202200511
Site-specific terminal and internal labeling of RNA by poly(A) polymerase tailing and copper-catalyzed or copper-free strain-promoted click chemistry. M.-L. Winz et al., 2012, Nucleic Acids Res., Vol. 40(10), p. e78. https://doi.org/10.1093/nar/gks062
Multiple covalent fluorescence labeling of eukaryotic mRNA at the poly(A) tail enhances translation and can be performed in living cells. L. Anhäuser et al., 2019, Nucleic Acids Res., Vol. 47(7), p. e42. https://doi.org/10.1093/nar/gkz084
Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides. C. W. Tornøe, C. Christensen, M. Meldal, 2002, J. Org. Chem., Vol. 67, p. 3057-3064. https://doi.org/10.1021/jo011148j
A Stepwise Huisgen Cycloaddition Process: Copper(I)-Catalyzed Regioselective “Ligation” of Azides and Terminal Alkynes. V. V. Rostovtsev, L. G. Green, V. V. Fokin, K. B. Sharpless, 2002, Angew. Chemie Int. Ed., Vol. 41, p. 2596–2599. https://doi.org/10.1002/1521-3773(20020715)41:14%3C2596::AID-ANIE2596%3E3.0.CO;2-4
FAQ
-
What is 2′-azido-2′-dATP used for?
It is primarily used for site-specific 3′-end labeling of nucleic acids via click chemistry or Staudinger ligation. It can also be used for co-transcriptional generation of internally azido-modified RNA.
-
Can this nucleotide be incorporated internally into Nucleic Acids?
Yes, it can also be used for co-transcriptional generation of internally azido-modified mRNA by IVT.
-
Which enzymes can incorporate 2′-azido-2′-dATP?
Commonly used enzymes include T7 RNA polymerase for internal RNA modification, and poly(A) polymerase or terminal deoxynucleotidyl transferase (TdT) for 3´-end modification of RNA.
-
Is it compatible with copper-free click chemistry?
Yes. The azido group supports SPAAC reactions with strained alkynes (e.g., DBCO) for copper-free labeling.
-
What are typical applications?
3´-end and internal mRNA modification for subsequent click conjugation with dyes, biotin, or ligands.

