
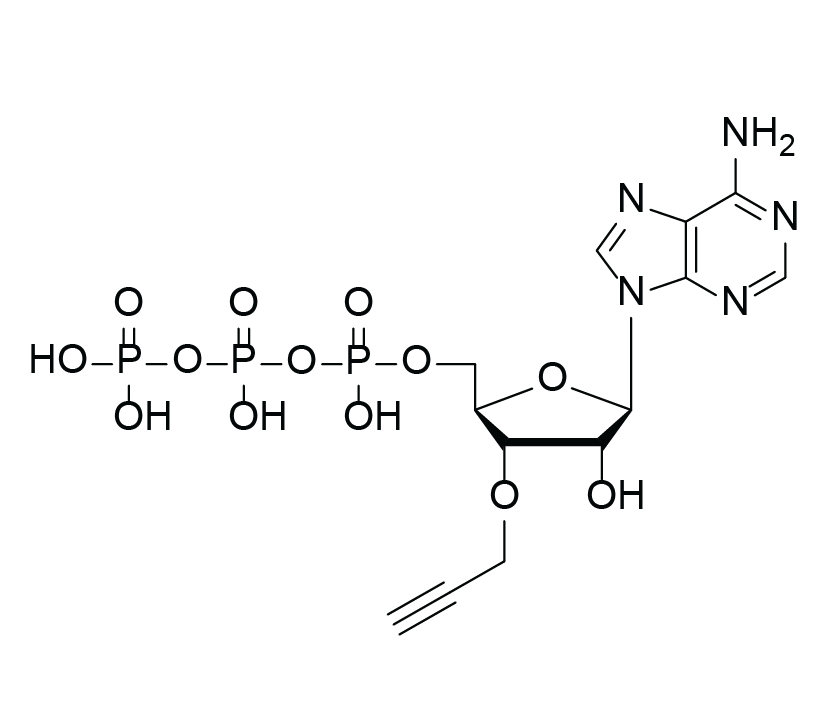
3′-(O-Propargyl)-ATP
Triphosphate for modifying of 3'END RNA
| Size | Catalog No. | Price |
|---|---|---|
| 1 µmol | BCT-31-S | € 800,00 |
Chemical Properties
-
Molecular Formula
C13H18N5O13P3 (free acid)
-
Shelf Life
12 months unopened after receipt
-
Storage Conditions
-20 °C, dark, dry
-
Molecular Weight
545.23 g/mol (free acid)
-
Purity
≥ 95% (HPLC)
-
Physical State
colorless to white solid
-
CAS Number
n.a.
-
Absorption (max)
λmax = 259 nm
-
Ɛ (max)
15.300 cm-1M-1
-
Salt Form
Triethylammonium salt
Product Information
Click‑Ready Triphosphate for 3′‑End RNA Labeling & Advanced mRNA Engineering
3′‑O‑Propargyl‑ATP is a high‑purity, alkyne‑modified adenosine triphosphate designed for site‑specific 3′‑end RNA modification. This nucleotide is incorporated by poly(A) polymerase, enabling precise installation of a terminal alkyne group on mRNA or other RNA species. Once incorporated, the alkyne allows highly selective and efficient CuAAC click chemistry with any azide‑functionalized label, fluorophores, biotin, ligands, DBCO adapters, and more.
As part of baseclick’s powerful portfolio of click‑enabled RNA nucleotides, 3′‑O‑Propargyl‑ATP plays a central role in mRNA R&D, RNA imaging, tracking studies, biodistribution assays, and targeted RNA conjugation strategies used in modern therapeutics.
Key Benefits 3′‑O‑Propargyl‑ATP
- Precise 3′‑End RNA Labeling
Enzymatically incorporated at the 3′‑end using poly(A) polymerase, ensuring single‑site alkyne installation.
- Universal Click‑Chemistry Compatibility
Once incorporated, the alkyne reacts cleanly with azide‑bearing dyes, tags, sugars, or drug‑delivery ligands using CuAAC chemistry.
- Flexible & Modular mRNA Functionalization
Avoids bulky dye‑labeled nucleotides by enabling post‑synthetic click labeling, offering absolute freedom in probe or ligand selection.
- High‑Purity Research Grade
≥ 95% HPLC purity ensures exceptional performance in advanced RNA workflows.
Applications of 3′‑O‑Propargyl‑ATP
3′‑O‑Propargyl‑ATP is designed for precise 3′‑end labeling of RNA using poly(A) polymerase, enabling the installation of a single terminal alkyne group on mRNA or other RNA molecules. This alkyne handle can then be functionalized through CuAAC click chemistry with azide‑modified dyes, biotin, ligands, or targeting molecules.
This makes the nucleotide ideal for a wide range of modern RNA research and engineering workflows, including:
Attach azide‑linked fluorophores for high‑resolution visualization in cells or in vivo.
- Targeted RNA conjugation
Click the 3′‑alkyne to sugars, peptides, or other ligands for investigating delivery pathways or receptor‑mediated uptake.
- Biodistribution & stability studies
Terminal tagging allows monitoring of mRNA persistence, decay, and intracellular routing.
- Modular post‑synthetic mRNA modification
Enables flexible introduction of imaging probes or functional groups without altering RNA sequence or IVT reaction conditions.
- Advanced mRNA engineering (incl. circular RNA workflows)
Serves as the required 3′‑alkyne handle for click‑mediated circularization or dual-end modification strategies.
FAQ
-
How is 3′‑O‑Propargyl‑ATP incorporated into RNA?
It is incorporated at the 3′‑end by poly(A) polymerase, producing RNA with a terminal alkyne for click chemistry.
-
Can it be used during in‑vitro transcription (IVT)?
No. It is specifically designed for post‑transcriptional 3′‑end modification, not for internal incorporation during IVT.
-
Which click chemistry is recommended?
Standard Cu(I)-catalyzed azide–alkyne cycloaddition (CuAAC) provides high yields and excellent specificity.
-
What can I attach to the alkyne?
Any azide‑bearing probe or ligand, including:
- Fluorescent dyes (FAM‑azide, TAMRA‑azide, AF647‑azide…)
- Biotin / desthiobiotin
- Sugar ligands for targeted delivery
- DBCO tags
- Peptides or small molecules
-
Why not use dye‑labeled ATP analogs instead?
Dye‑ATP analogs are often large and poorly accepted by polymerases.
3′‑O‑Propargyl‑ATP offers clean enzymatic incorporation followed by flexible, post‑synthetic labeling with any azide probe.

