
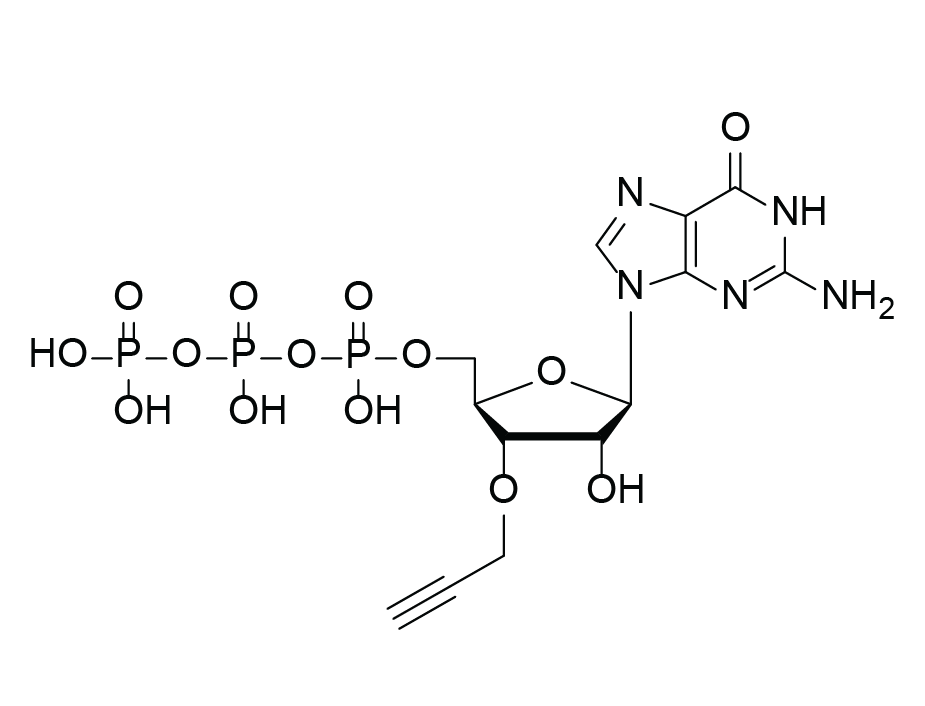
3′-(O-Propargyl)-GTP
Triphosphate for cap modifying of 5'END RNA
| Size | Catalog No. | Price |
|---|---|---|
| 1 µmol | BCT-32-S | € 800,00 |
Chemical Properties
-
Molecular Formula
C13H18N5O14P3 (free acid)
-
Shelf Life
12 months unopened after receipt
-
Storage Conditions
-20 °C, dark, dry
-
Molecular Weight
561.23 g/mol (free acid)
-
Purity
≥ 95% (HPLC)
-
Physical State
colorless to white solid
-
CAS Number
1610778-63-6
-
Absorption (max)
λmax = 252 nm
-
Ɛ (max)
13.700 cm-1M-1
-
Salt Form
Triethylammonium salt
Product Information
Triphosphate for 5′‑End RNA Modification and Click‑Compatible Capping Strategies
3′‑O‑Propargyl‑GTP is a guanosine triphosphate analog carrying a terminal alkyne group at the 3′‑O position of the ribose. This modification enables the introduction of a click‑reactive alkyne at the 5′ end of short or synthetic RNAs meanwhile forming a protective cap structure, using capping enzyme–based approaches that creates a GpppN structure capable of supporting efficient biological activity.
Although 3′‑O‑Propargyl‑GTP can be incorporated enzymatically at the 3′‑end of RNA, its primary application is in 5′‑end engineering, where it is added by a capping enzyme and subsequently methylated. This results in a cap‑mimicking 5′ modification that maintains translational or functional integrity while enabling downstream CuAAC click conjugation with azido‑labeled probes.
Key Features
- Reactive Alkyne at the 5′ Terminus
Provides a clickable handle directly at the RNA 5′ end cap for CuAAC‑based conjugation with azide dyes, ligands, biotin, or functional groups.
- Cap‑Like Structure for Functional Short RNA Constructs
The enzymatically incorporated and methylated 3′‑O‑Propargyl‑GTP forms a cap‑like 5′ modification, suitable for functionalized small RNAs, synthetic guide RNAs, or engineered RNA reporters.
- High‑Purity Research Grade
≥ 95% purity ensures reliable performance in RNA modification workflows.
- Ideal for Short RNA and Synthetic RNA Applications
Supports experimental systems requiring functionalized RNAs independent of full‑length mRNA processes.
Applications
- 5′‑End Modification of Short or Synthetic RNA
3′‑O‑Propargyl‑GTP can be added enzymatically to mimic a RNA 5′ cap by forming the GpppN structure, introducing an alkyne for precise chemical modification. This is well suited for short RNAs, including functional RNAs, labeled guide RNAs, aptamers, or reporter RNAs.
- Click‑Chemistry Conjugation at the 5′ Position
The terminal alkyne enables highly selective CuAAC reactions to attach azide‑labeled dyes, affinity tags, peptides, or bioactive molecules. This approach provides a clean and modular way to build custom 5′‑functionalized RNA tools.
- Construction of Cap‑Like Structures for Functional RNA
Using a capping enzyme, 3′‑O‑Propargyl‑GTP can be incorporated at the RNA 5′ end and then methylated to create a cap‑like structure that maintains compatibility with processes requiring capped or cap‑mimicking RNA ends.
- Fluorescent or Biochemical RNA Labeling
The clickable 5′ alkyne allows selective introduction of imaging probes or chemical handles, enabling sensitive detection of short RNAs in biochemical assays.
FAQ
-
Can 3′‑O‑Propargyl‑GTP be used like 3′‑O‑Propargyl‑ATP for 3′‑end labeling?
It can be incorporated at the 3′‑end, but 3′‑O‑Propargyl‑ATP (BCT‑31) is more efficient for tail‑labeling, as confirmed by internal comparison on the product page.
-
What makes 3′‑O‑Propargyl‑GTP unique?
Its strength lies in 5′‑end modification via capping enzymes, providing a cap‑like, functionalizable structure not achievable with standard nucleotide triphosphates.
-
Is 3′‑O‑Propargyl‑GTP compatible with click chemistry?
Yes, its alkyne moiety is fully compatible with CuAAC labeling using azide-functionalized molecules.

