
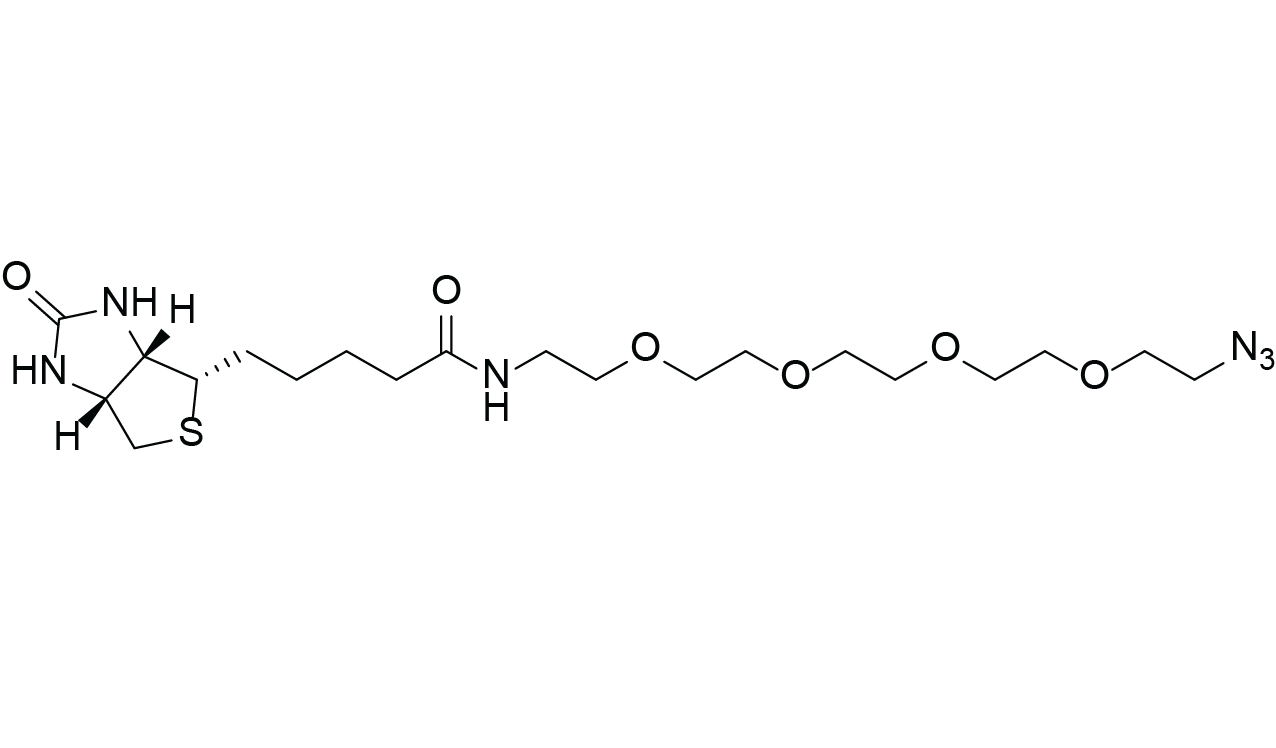
Biotin-PEG4-Azide
Biotinylation reagent for labeling by CuAAC
| Size | Catalog No. | Price |
|---|---|---|
| 5 mg | BCFA-234-5 | € 70,00 |
| 10 mg | BCFA-234-10 | € 110,00 |
Chemical Properties
-
Molecular Formula
C20H36N6O6S
-
Shelf Life
12 months unopened after receipt
-
Storage Conditions
-20 °C, dark
-
Molecular Weight
488.6 g/mol
-
Purity
≥ 95% (HPLC)
-
Physical State
white to light yellow solid to lump
-
CAS Number
1309649-57-7
-
Solubility
DMSO, DMF
-
Preparation/Handling
For a 10 mM solution add 1023 μL to 5 mg.
For a 10 mM solution add 2047 μL to 10 mg.
Product Information
A biotin labeling reagent for biomolecules using azide alkyne cycloaddition (Click Chemistry)
What is biotin
Biotin, also known as vitamin B7, is a sulfur and nitrogen containing bicyclic heterocycle that functions as an essential coenzyme in cellular metabolism. It is required for carboxylation reactions catalyzed by enzymes such as acetyl CoA carboxylase and pyruvate carboxylase.
Beyond its biological role, biotin is renowned for forming one of the strongest known non covalent interactions with the proteins avidin and streptavidin. This exceptionally stable interaction is widely exploited in modern biochemical research, particularly in peptide, protein, and nucleic acid applications.
Applications of biotin
Peptide/protein research:
The biotin–streptavidin (or avidin) interaction underpins numerous techniques in peptide and protein research. Biotinylation of peptides and proteins is widely used for affinity purification, as biotin labeled targets can be captured on streptavidin or avidin coated magnetic beads, resins, or columns, enabling rapid one step purification.
In addition, biotin labeling allows sensitive detection of proteins through streptavidin conjugated to reporter molecules, most commonly fluorophores or enzymes such as horseradish peroxidase (HRP). Consequently, biotinylation is frequently employed in immunoassays including ELISA, Western blotting, and immunohistochemistry.
Biotin tagging also supports the enrichment of specific protein populations, such as newly synthesized proteins, for downstream mass spectrometry analysis. Furthermore, immobilization of biotinylated proteins on streptavidin coated surfaces enables binding assays to study protein–protein interactions.
Nucleic acid research:
In nucleic acid research, biotin labeling is primarily used for purification and capture applications. The strong interaction between biotin and streptavidin or avidin allows biotinylated DNA or RNA to be immobilized on beads, columns, or resins, facilitating efficient separation from impurities.
Biotin containing probes are commonly applied in in situ hybridization (ISH), Southern blotting, and Northern blotting to target specific DNA or RNA sequences. After hybridization, these probes can be visualized and localized using streptavidin or avidin based detection reagents, typically conjugated to fluorophores or enzymes.
Biotin Labeling via Click Chemistry
All of the above applications require reliable production of biotin labeled biomolecules. Click chemistry offers a simple, cost effective, and highly specific approach to achieve this. For click based labeling, the target biomolecule must carry a terminal alkyne or a strained alkyne functionality.
Strained alkynes enable catalyst free reactions and are especially suitable for sensitive biological applications. Common examples include DBCO and BCN groups. Biomolecules containing these modifications are widely available. baseclick offers biotin labeled oligonucleotides and mRNA as well as custom synthesis of alkyne or DBCO modified nucleic acids for flexible downstream labeling. For other targets, support is available via support@baseclick.eu.
Chemical Properties of Biotin-PEG4-Azide
Biotin-PEG4-Azide is a versatile reagent for biotin labeling under mild conditions. Covalent conjugation to target molecules can be achieved via two click chemistry-based reactions:
- Strain promoted azide–alkyne cycloaddition (SPAAC): A copper free reaction requiring neither heat nor catalyst, resulting in a stable triazole linkage with strained alkynes.
- Copper catalyzed azide–alkyne cycloaddition (CuAAC): A selective reaction forming a 1,4 substituted triazole linkage between biotin and the target molecule.
Both reactions are considered bioorthogonal, meaning they proceed without interfering with native biological functionalities. The hydrophilic PEG4 spacer reduces steric hindrance between the biotin and the labeled biomolecule while significantly increasing water solubility.
These properties make Biotin-PEG4-Azide an ideal choice for biotin labeling of a wide range of alkyne modified biomolecules, including peptides, proteins, and nucleic acids. The option to use copper free SPAAC click chemistry further enables in vivo labeling applications.
Because click chemistry relies on non-naturally occurring functional groups, the site of biotin attachment can be precisely controlled, in contrast to traditional labeling approaches such as NHS ester based biotinylation.
LITERATURE
Discovery of an NAD+ analogue with enhanced specificity for PARP1, X. N. Zhang et al., 2022, Chemical Science, Vol. 13(7), p. 1982-1991.
https://doi.org/10.1039/D1SC06256E
Epitopes displayed in a cyclic peptide scaffold bind SARS‐CoV‐2 antibodies, C. Eriksson et al., 2023, ChemBioChem, Vol. 24(15), e202300103.
https://doi.org/10.1002/cbic.202300103
Functionalized scout fragments for site-specific covalent ligand discovery and optimization, V. M. Crowley et al., 2021, ACS Central Science, Vol. 7(4), p. 613-623.
https://doi.org/10.1021/acscentsci.0c01336
FAQ
-
What is Biotin‑PEG4‑Azide used for?
Biotin‑PEG4‑Azide is a versatile reagent for biotinylating biomolecules using azide–alkyne click chemistry, including both SPAAC (copper‑free) and CuAAC reactions. It enables clean, efficient, and bioorthogonal biotin labeling of peptides, proteins, nucleic acids, and other alkyne‑modified targets.
-
What are the advantages of the PEG4 spacer?
The PEG4 hydrophilic linker:
- reduces steric hindrance between biotin and the target molecule
- increases water solubility
- minimizes nonspecific interactions
- improves accessibility to streptavidin during detection or purification
This results in higher reaction efficiency and better performance in complex biological environments.
-
Can Biotin‑PEG4‑Azide be used for copper‑free click chemistry (SPAAC)?
Yes. Biotin‑PEG4‑Azide is fully compatible with strain‑promoted azide–alkyne cycloaddition and reacts efficiently with strained alkynes such as DBCO or BCN—ideal for live cells, sensitive proteins, and in vivo applications where copper must be avoided.
-
Can it also be used in copper‑catalyzed click chemistry (CuAAC)?
Yes. Biotin‑PEG4‑Azide forms highly stable, selective 1,4‑triazole linkages with terminal alkynes under standard CuAAC conditions. Both SPAAC and CuAAC reactions yield robust and bioorthogonal conjugates.
-
Which biomolecules can be labeled using Biotin‑PEG4‑Azide?
It is suitable for a wide range of alkyne‑ or strained‑alkyne‑modified targets, including:
- peptides and proteins
- DNA and RNA (including oligos and mRNA)
- polysaccharides
- viruses and viral particles
- surface‑presented biomolecules on beads or nanoparticles
baseclick also provides biotinylated oligos/mRNA and custom alkyne/DBCO modifications.
-
Why is biotin widely used in biochemical research?
Biotin forms one of the strongest known non‑covalent interactions with streptavidin and avidin, enabling:
- affinity purification
- high‑sensitivity detection in ELISA, Western blot, and IHC
- immobilization of biomolecules on streptavidin surfaces
- enrichment of newly synthesized proteins
- nucleic acid detection in ISH, Southern, and Northern blots
Streptavidin is generally preferred due to better solubility and lower nonspecific binding.
-
Why choose Biotin‑PEG4‑Azide over NHS‑biotin?
Click‑based biotinylation allows:
- precise, site‑specific labeling only at alkyne sites
- compatibility with sensitive proteins and nucleic acids
- no reaction with lysines or other amines
- avoidance of over‑labeling or loss of biological function
- NHS‑ester biotinylation is less selective and typically labels multiple sites.
-
What are the typical applications of Biotin‑PEG4‑Azide?
Common uses include:
- affinity purification of proteins or nucleic acids
- labeling for fluorescence or enzymatic detection
- immobilization of biomolecules on streptavidin‑coated plates, surfaces, or beads
- diagnostic assay development
- tracking protein–protein interactions
- nucleic‑acid hybridization workflows
- live‑cell or in vivo labeling via SPAAC
-
What are the chemical strengths of Biotin‑PEG4‑Azide?
Key properties include:
- copper‑free and copper‑catalyzed click compatibility
- high aqueous solubility
- low steric hindrance via PEG4
- stable triazole linkages
- bioorthogonal reactivity with no interference in biological systems
-
How should Biotin‑PEG4‑Azide be stored?
Store at –20 °C, protected from light and moisture. Avoid repeated freeze–thaw cycles to maintain reagent integrity.
-
Does baseclick offer related or complementary products?
Yes. baseclick provides a broad toolbox including:
- Biotin‑Azide Plus (for low‑copper CuAAC)
- Biotin‑DBCO reagents
- DBCO‑linkers for SPAAC
- Click‑compatible fluorophores (FAM, TAMRA, Cy5, etc.)
Modified oligos and mRNA with alkyne or DBCO groups.
-
Can baseclick support custom biotinylation workflows?
Absolutely. baseclick offers custom synthesis of:
- alkyne‑modified oligonucleotides
- DBCO‑modified nucleic acids
- biotinylated mRNA
- tailored conjugation strategies
Contact support@baseclick.eu for personalized assistance.

