
CleanCap
Cap structure for protection of mRNA
| Size | Catalog No. | Price |
|---|---|---|
| 1 µmol | BCT-38-S | € 350,00 |
Chemical Properties
-
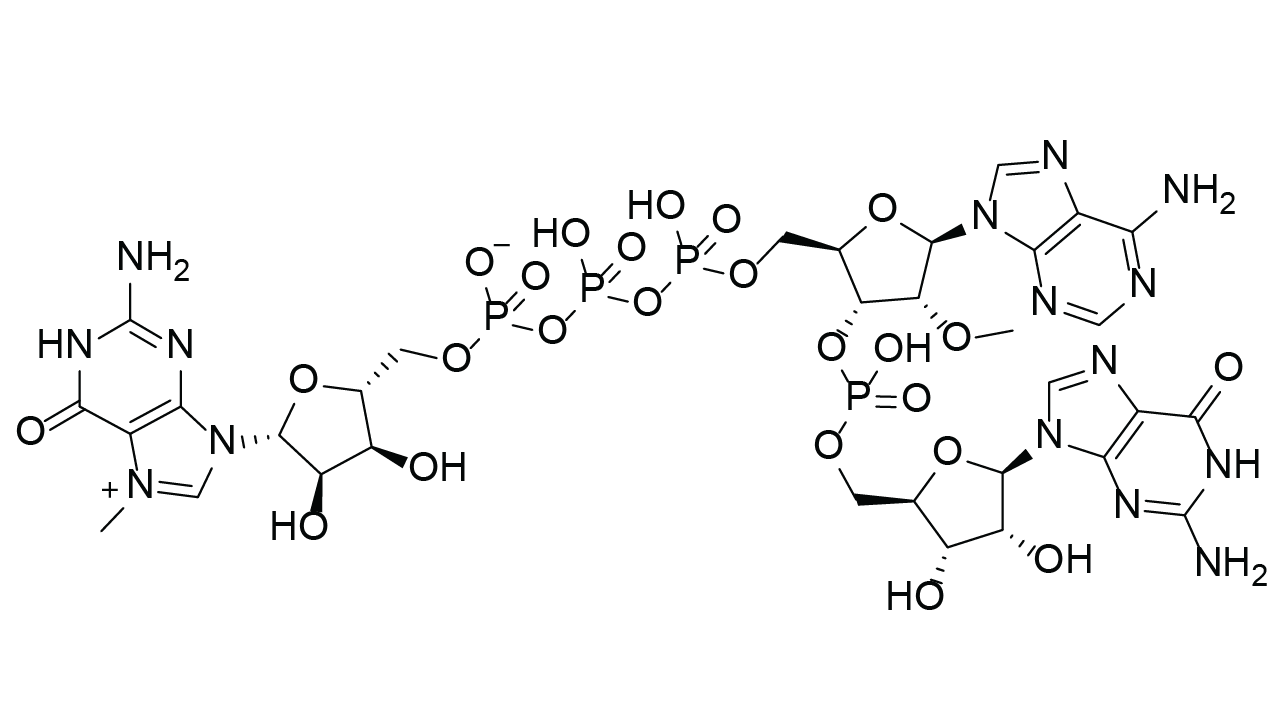
Molecular Formula
C32H43N15O24P4 (free acid)
-
Shelf Life
12 months unopened after receipt
-
Storage Conditions
-20 °C, dark, dry
-
Molecular Weight
1145.60 g/mol (free acid)
-
Purity
≥ 95% (HPLC)
-
Physical State
colorless solution (100 ± 6 mM)
-
CAS Number
n.a.
-
Absorption (max)
λmax = 255 nm
-
Ɛ (max)
30,539 cm-1M-1
-
Additional name
m7G(5′)ppp(5′)(2’OMeA)pG
Product Information
A Next‑Generation High‑Efficiency mRNA Capping Technology
CleanCap is a next‑generation co‑transcriptional mRNA capping technology designed to produce a naturally occurring Cap1 mRNA 5´end during in vitro transcription (IVT). It is a trinucleotide cap analog that directs T7 RNA polymerase to initiate transcription in a precise, sequence‑dependent manner, resulting in exceptionally high capping efficiency and biologically authentic mRNA for efficient translation.
CleanCap AG provides high-quality capped transcripts with minimal byproducts. This high efficiency is possible because it aligns with a defined 5’ initiating sequence of the DNA template that must start with AG, ensuring accurate incorporation of CleanCap AG at the 5´end of the growing RNA strand.
The CleanCap approach produces Cap 1 structures, the dominant form of capped mRNA in higher eukaryotes, which enhance mRNA stability, support efficient translation initiation, and reduce recognition by innate immune sensors. This makes CleanCap‑generated mRNA more physiologically compatible and better suited for therapeutic applications.
CleanCap AG is widely used in the production of mRNA transcripts for vaccines, gene therapies, and research applications. The CleanCap technology is simple, one‑pot, and compatible with standard IVT reactions.
Key Benefits of CleanCap AG
- Natural Cap1 structure for improved stability and translation.
- ≥ 95% capping efficiency for high‑purity, high‑performance mRNA.
- One‑pot IVT integration for faster, simplified mRNA production.
- High transcription yields (4–5 mg/mL) suitable for scale‑up.
- Low innate immune activation thanks to biologically accurate design.
Applications
- mRNA vaccines requiring high expression and low immunogenicity
- Gene therapy and ex vivo cell engineering
- High‑yield IVT manufacturing platforms
- Research mRNA for protein expression studies
- mRNA stability and delivery optimization
Note: CleanCap® is a registered trademark of TriLink BioTechnologies. CleanCap® technology is patented by TriLink BioTechnologies. baseclick offers CleanCap® products exclusively for research applications.
LITERATURE
Cap 1 Messenger RNA Synthesis with Co-transcriptional CleanCap® Analog by In Vitro Transcription. M. Henderson et al., 2021, Curr. Protoc., Vol. 1, p. e39.
https://doi.org/10.1002/cpz1.39
Revolutionizing immunization: a comprehensive review of mRNA vaccine technology and applications. K. Y. Leong et al., 2025, J., Vol. 22, 71.
https://doi.org/10.1186/s12985-025-02645-6
Development of bis-ANS-based modified fluorescence titration assay for IFIT/RNA studies, Anna Dobieżyńska et al., 2020, ScienceDirect, Vol. 553(3), p. 391-396.
https://doi.org/10.1016/j.bbrc.2020.09.006
Enhanced prime editing systems by manipulating cellular determinants of editing outcomes, Peter J. Chen et al., 2021, Cell, Vol. 184(22), p. 5635-5652.
https://doi.org/10.1016/j.cell.2021.09.018
Added to pre-existing inflammation, mRNA-lipid nanoparticles induce inflammation exacerbation (IE), Hamideh Parhiz et al., 2022, Journal of Controlled Release, Vol. 344, p. 50-61.
https://doi.org/10.1016/j.jconrel.2021.12.027
FAQ
-
What makes CleanCap different from older capping methods?
CleanCap introduces the Cap 1 structure during transcription, eliminating the need for extra enzymatic capping steps and providing much higher incorporation efficiency (≥ 95% Cap1) and higher IVT yields (up to ~5 mg/mL).
-
What is a Cap1 structure, and why does it matter?
Cap1 is the naturally occurring mRNA cap in higher eukaryotes. It enhances:
- mRNA stability
- translational efficiency
- and also reduces innate immune activation
CleanCap® produces Cap1 as the default output.
-
Is CleanCap® available in GMP grade?
Yes. CleanCap is manufactured in accordance with GMP guidelines, is used in multiple commercial vaccine programmes and is available for use in preclinical studies through to clinical applications.

