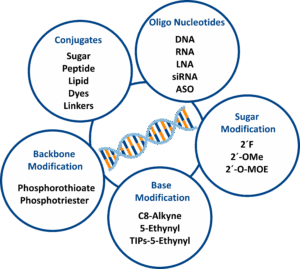
Oligonucleotide synthesis is the precise and controlled process of creating short DNA or RNA sequences that serve as essential tools in research, diagnostics, and therapeutic development. These synthetic strands form the backbone of modern molecular biology, enabling applications such as PCR, hybridization assays, antisense technologies, sequencing methods and RNA-based therapeutics. The ability to customize oligonucleotides with chemical modifications has transformed their functionality, allowing scientists to design molecules that not only hybridize with target sequences but also carry additional features for detection, delivery, or enhanced stability.
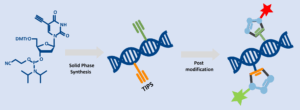
The cornerstone of oligonucleotide production is solid-phase chemical synthesis, a highly reliable method that assembles nucleotides stepwise on a solid support. This technique offers exceptional precision and flexibility, making it possible to introduce a wide range of modifications during synthesis. Among these are click-reactive handles, which enable post-synthetic conjugation with dyes, ligands, sugars, and other biomolecules. Such modularity is critical for creating advanced probes, targeted delivery systems, and multifunctional therapeutic constructs.
While enzymatic synthesis can produce very long sequences often exceeding 200 nucleotides it is rarely required for standard oligonucleotide applications such as short probes or antisense molecules. Instead, enzymatic approaches are typically reserved for specialized cases like mRNA production or specific next-generation sequencing workflows. For most research and diagnostic needs, solid-phase synthesis remains the preferred method because it supports precise sequence control and complex chemical modifications at the 5′ end, 3′ end, and internal positions.
At baseclick, solid-phase synthesis of oligonucleotides, a service provided by metabion, is combined with click chemistry to deliver high-purity, customizable oligonucleotides that meet the demands of modern science. This integrated approach ensures scalability, reproducibility, and compatibility with advanced applications, from RNA therapeutics to functional probes for imaging and detection.
Solid‑Phase Chemical Synthesis
Solid‑phase synthesis is the established technique for constructing oligonucleotides through a controlled, stepwise process, during which the growing strand remains anchored to solid support. This approach enables tight control of sequence, length, and chemical environment.
The synthesis proceeds from the 3′ end toward the 5′ end, using phosphoramidite chemistry. Each cycle includes deprotection, coupling, capping of unreacted chains, and oxidation to stabilize the phosphodiester bond.
Solid-phase synthesis enables the incorporation of a wide range of chemical modifications, including dyes, functional building blocks, and sugar-based conjugation motifs. Although internal alkyne phosphoramidites (such as alkyne-dC and alkyne-dT) are available for precise position-specific functionalization within the oligonucleotide sequence, in practice, modifications are still performed almost exclusively at the terminal ends. Terminal functionalization offers high reaction efficiency, simplifies purification, and meets the requirements of most diagnostic and therapeutic applications.
Solid‑phase synthesis facilitates clean purification and high reproducibility, essential for therapeutic‑grade oligos and multi‑label probes.
Enzymatic Oligonucleotide Synthesis
Enzymatic synthesis uses polymerases and nucleoside triphosphates (NTPs or dNTPs) to produce DNA or RNA molecules under biological conditions. This method is particularly valuable when generating long RNA transcripts such as mRNA or for introducing modifications via engineered nucleotides. It also complements chemical synthesis by enabling smooth workflows for RNA therapeutics, mRNA stabilization strategies, and click‑enabled NGS library preparation technologies such as ClickSeq.
Enzymatic methods allow the incorporation of modified triphosphates, including those that enhance stability, reduce immunogenicity (such as 5´‑Ethynyluridine, or introduce click‑reactive functionalities directly during polymerization. This makes enzymatic synthesis both biologically relevant and chemically flexible.
Click Chemistry in Oligonucleotide Engineering
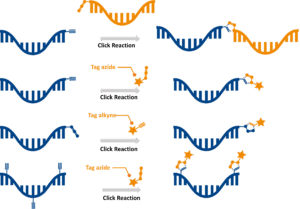
Click chemistry refers to a set of high‑efficiency, bioorthogonal reactions enabling covalent linkage between azide‑ and alkyne‑functionalized molecules. In oligonucleotide engineering, click chemistry provides a robust, reproducible way to attach functional ligands as dyes, lipids, sugar clusters, or affinity tags.
Common reactions supported by baseclick include:
- CuAAC (Copper‑catalyzed Azide–Alkyne Cycloaddition): high‑yield, versatile, ideal for late‑stage functionalization when copper is tolerated.
- SPAAC (Strain‑Promoted Azide–Alkyne Cycloaddition): copper‑free and suitable for in vivo or copper‑sensitive conjugations.
Click chemistry is central to baseclick’s modular conjugation platform, enabling late‑stage, site‑specific functionalization without compromising oligo integrity.
5′‑End Modifications
The 5′ terminus is a strategic location for attaching functional groups because modifications here do not interfere with base‑pairing. Through chemical synthesis or post‑synthetic click reactions, the 5′ end can be functionalized for imaging, therapeutic delivery, or diagnostic signal amplification.
Typical 5′ modifications include:
- 5′‑azide or 5′‑alkyne handles for CuAAC or SPAAC conjugation.
- Fluorophores and dyes used in imaging, qPCR, or FISH assays.
- Biotin and biotin analogs for affinity capture or detection systems.
- Sugar ligands such as GalNAc, mannose, tri‑mannose, or tr‑GalNAc, which mediate highly selective uptake in hepatocytes or macrophages, depending on ligand type.
These conjugates enable powerful targeted delivery platforms and diagnostic tools.
3′‑End Modifications
The 3′ terminus is often modified to enhance nuclease resistance, extend RNA or mRNA stability, and control biological half‑life. Modifications at this site can protect the molecule from exonucleases and improve its biochemical performance, which is highly relevant for therapeutic oligonucleotides.
3′ modifications may include:
- Dye labels for visualization and tracking.
- Click‑reactive groups enabling conjugation with ligands or functional moieties.
- Sugar‑based ligands for targeted delivery.
- Quenchers for qPCR (hydrolysis) probes
3′ labeling can stabilize mRNA, reduce immune activation, and support longer protein expression, making 3′ modifications essential in therapeutic and vaccine development workflows.
Internal Modifications
Internal modifications introduce functional elements within the oligo sequence rather than at a terminus. Such modifications are critical for applications requiring multi‑point labeling, fluorescence resonance energy transfer (FRET), multi‑valent ligand display, or structural probing.
baseclick’s internal alkyne phosphoramidites, together with metabions experience in custom oligonucleotide synthesis, enable:
- Precision placement of clickable handles within the sequence.
- Multi‑site conjugation for dual‑color imaging or multi‑ligand architectures.
- High labeling efficiency, even when multiple clickable units are present.
- Dual-Click Strategy: combines TIPS-5-Ethynyl-dU-CEP with for example C8-Alkyne-dU-CE Phosphoramidite in the same oligo to create two independent clickable sites:
Backbone and sugar chemistry adjustments including phosphorothioate (PS) linkages, 2′‑OMe, 2′‑MOE, 2′‑F, LNA and cEt—further tune stability, potency, and pharmacokinetics, especially in antisense and siRNA therapeutics. These are fully compatible with click‑based conjugation strategies.