
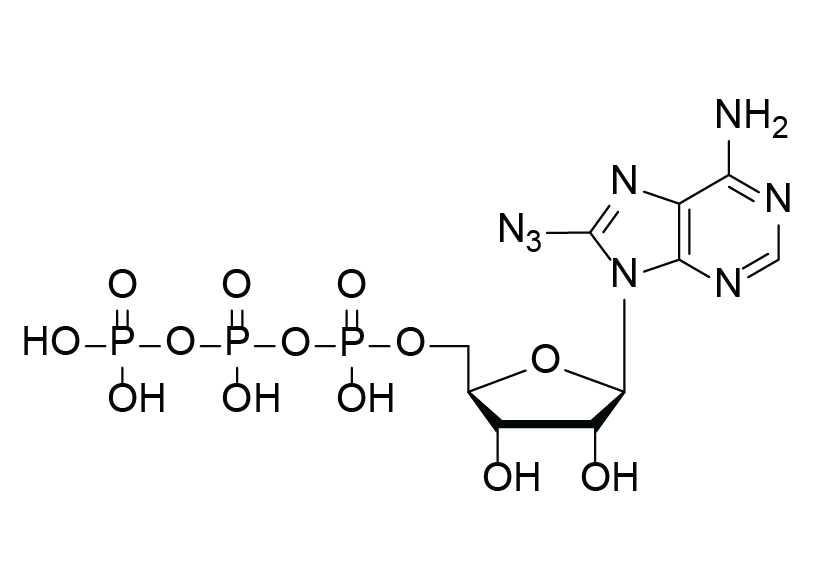
8-Azido-ATP
Triphosphate for modifying of 3'END RNA
| Size | Catalog No. | Price |
|---|---|---|
| 1 µmol | BCT-33-S | € 90,00 |
| 5 µmol | BCT-33-L | € 240,00 |
Chemical Properties
-
Molecular Formula
C10H17N8O13P3
-
Shelf Life
12 months unopened after receipt
-
Storage Conditions
-20 °C, dark, dry
-
Molecular Weight
548.19 g/mol (free acid)
-
Purity
≥ 95% (HPLC)
-
Physical State
colorless to slightly yellow solution (10 mM)
-
CAS Number
53696-59-6
-
Absorption (max)
λmax = 281 nm
-
Ɛ (max)
13.300 cm-1M-1
Product Information
A Nucleotide Analogue for Photoaffinity-Labeling of ATP-Dependent Proteins
Molecular Structure and Applications of 8-Azido-ATP
8-Azido-adenosine 5´-triphosphate (8-Azido-ATP) is a chemically modified analogue of adenosine triphosphate (ATP) with an azido (N₃) group at the position 8 of the adenine base.
This modification enables selective photoreactivity and biorthogonal conjugation chemistry while maintaining compatibility with ATP-binding proteins and certain enzymatic processes. Upon UV radiation, the azido group converts into a highly reactive nitrene species, enabling covalent crosslinking to ATP-depend proteins for photoaffinity labeling.[1-4]
Additionally, 8-Azido-ATP is suited for the site-specific, post-synthetic introduction of azido groups at the 3′ end of nucleic acids, using enzymes such as poly(A) polymerase. The azido group enables the 3′ end to be modified with fluorescent dyes, biotins, linkers, or cell-targeting ligands via click chemistry.
The azido group: Photoaffinity Labeling of Proteins and Biorthogonal RNA Labeling via Click Chemistry
The azido group is a biorthogonal functional group, meaning that it does not interfere with natural biomolecules. Upon exposure to UV light, the azido group is converted into a reactive nitrene group which covalently links the molecule to its protein binding partner.
The azido group also allows a highly efficient ligation of fluorescent dyes, biotins, linkers, or cell-targeting ligands to nucleic acids by click chemistry.[5, 6] Here, the azido group reacts selectively with:
- alkynes, forming triazoles via copper-catalysed azide–alkyne cycloaddition (CuAAC) or copper-free strain-promoted azide–alkyne cycloaddition (SPAAC).
- phosphines, forming amides via Staudinger ligation.
The development of the azide–alkyne cycloaddition was recognized with the Nobel Prize in Chemistry in 2022.[5, 6]
Application Areas for 8-Azido-ATP
- Photoaffinity Labeling of ATP-Dependent Proteins: upon exposure to UV light, the azido group is converted into a reactive nitrene group which covalently links the molecule to its protein binding partner.[1-4]
- 3′-End Labeling of Nucleic Acids: post-transcriptional enzymatic incorporation / addition to the 3´-end, followed by click conjugation with dyes, biotin, or ligands.[5, 6]
- Advanced Bioconjugation: allows the attachment of sugars or targeting moieties for active mRNA transport into cells.
- Copper-Free Click Chemistry In Vivo: the azido group of 8-azido-ATP is a biorthogonal functionality, meaning that it does not interfere with natural biomolecules.
- Compatible with Enzymatic Incorporation: 8-azido-ATP is accepted as substrate by poly(A) polymerase.
Protocols and Resources
baseclick provides detailed protocols for click conjugation with fluorescent dyes, biotin, linkers, and targeting ligands.
LITERATURE
[1] Direct Photoaffinity Labeling of the Kir6.2 Subunit of the ATP-sensitive K+ Channel by 8-Azido-ATP. K. Tanabe et al., 2019, J. Biol. Chem., Vol. 274(7), p. 3931-3933. https://www.jbc.org/article/S0021-9258(19)87862-6/fulltext
[2] ATP binding and hydrolysis properties of ABCB10 and their regulation by glutathione. W. Qiu et al., 2015, PLoS One, Vol. 10(6), e0129772. https://doi.org/10.1371/journal.pone.0129772
[3] Identification of a plant receptor for extracellular ATP. J. Choi et al., 2014, Science, Vol. 343(6168), p. 290-294. https://doi.org/10.1126/science.343.6168.290
[4] ATP binding to two sites is necessary for dimerization of nucleotide-binding domains of ABC proteins. M. E. Zoghbi et al., 2014, Biochem. Biophys. Res. Commun., Vol. 443(1), p. 97-102. https://doi.org/10.1016/j.bbrc.2013.11.050
[5] Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides. C. W. Tornøe, C. Christensen, M. Meldal, 2002, J. Org. Chem., Vol. 67, p. 3057-3064. https://doi.org/10.1021/jo011148j
[6] A Stepwise Huisgen Cycloaddition Process: Copper(I)-Catalyzed Regioselective “Ligation” of Azides and Terminal Alkynes. V. V. Rostovtsev, L. G. Green, V. V. Fokin, K. B. Sharpless, 2002, Angew. Chemie Int. Ed., Vol. 41, p. 2596–2599. https://doi.org/10.1002/1521-3773(20020715)41:14%3C2596::AID-ANIE2596%3E3.0.CO;2-4
FAQ
-
What is 8-Azido-ATP used for?
It is primarily used for photoaffinity labeling of ATP-dependent proteins.[1-4]
It can also be used for site-specific 3′-end labeling of nucleic acids via click chemistry or Staudinger ligation.[5, 6]
-
Which enzymes can incorporate 8-Azido-ATP?
Commonly poly(A) polymerase is used for 3´-end modification of RNA with this modification.
-
Is it compatible with copper-free click chemistry?
Yes. The azido group supports SPAAC reactions with strained alkynes (e.g., DBCO) for copper-free labeling.
-
What are typical applications?
Photoaffinity labeling of ATP-dependent proteins or site-specific 3′-end labeling of nucleic acids via click chemistry.[1-4]

